Lidocaine/Prilocaine
$60.00 $50.00
lidocaine and prilocaine cream | lidocaine prilocaine cream
Lidocaine/prilocaine is a eutectic mixture of equal quantities (by weight) of lidocaine and prilocaine. lidocaine and prilocaine cream
A 5% emulsion preparation, containing 2.5% each of lidocaine/prilocaine, is under the trade name EMLA (an abbreviation for Eutectic Mixture of Local Anesthetics).
As a spray, under the name Tempe (topical eutectic-like mixture for premature ejaculation) (PSD502)
that can utilise by men to help with premature ejaculation.
The spray is produce by Plethora Solutions and branded as Fortacin in the UK and Recordati in the EU.
Clinical use of Lidocaine/Prilocaine | lidocaine prilocaine | lidocaine 2.5 and | | prilocaine 2.5 cream | lidocaine and prilocaine | lidocaine prilocaine cream uses
Indications | lidocaine and prilocaine cream
The lidocaine/prilocaine combination is indicated for dermal anaesthesia.
Specifically it is apply to prevent pain with intravenous catheter insertion,
blood sampling, superficial surgical procedures, and topical anaesthesia of leg ulcers for cleansing or debridement.
Also, it can be used to numb the skin before tattooing as well as electrolysis and laser hair removal.
It is also sometimes used in advance of injected local anaesthetics for minor surgery and biopsies.
A topical spray consisting of an aerosol formulation of lidocaine and
prilocaine was evaluated under the name PSD502 for use in treating premature ejaculation.
The spray is on the penile skin prior to intercourse.
While this formulation was not approved by the FDA, a similar product, Promescent, is available over-the-counter in the U.S.
Dosage forms
Lidocaine/prilocaine eutectic mixture is market as a 5% oil-in-water emulsion incorporate in a cream base (EMLA cream) or a cellulose disk (EMLA patch).
The cream is apply under an occlusive dressing, while the patch incorporates an occlusive dressing to facilitate absorption
of lidocaine and prilocaine into the area where anaesthesia is require.
Local dermal anaesthesia is achieve after approximately 60 minutes, whereupon the occlusive dressing (or patch) is remove.
The duration of anaesthesia is approximately two hours following removal of the occlusive dressing.
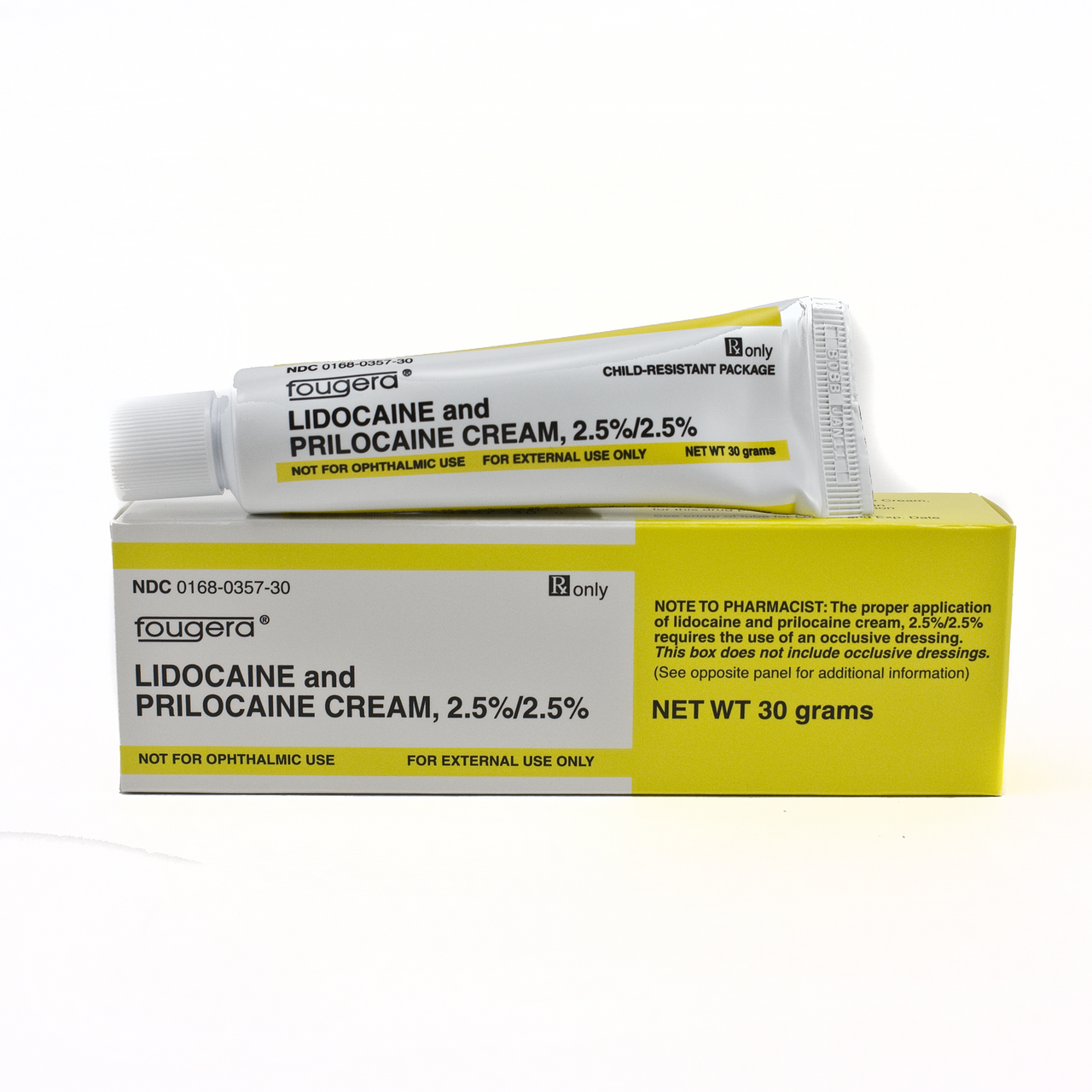
E. Fougera & Co., makers of the generic cream widely used in the United States as Lidocaine and Prilocaine Cream, 2.5%/2.5%, recommends different timing for application of the cream as well as length of anesthesia.
Additionally, they state that the duration of effective skin anesthesia will be at least one hour after removal of the occlusive dressing.
-
lidocaine and prilocaine cream price,
-
lidocaine and prilocaine spray,
-
lidocaÃna/prilocaÃna,
-
prilocaine and lidocaine,
-
what is lidocaine 2.5 and prilocaine 2.5 cream used for,
-
2.5 lidocaine 2.5 prilocaine,
-
akorn lidocaine 2.5 and prilocaine 2.5 cream,
-
akorn lidocaine and prilocaine cream,
-
emla lidocaine prilocaine,
-
eutectic mixture of lidocaine and prilocaine,
-
how to apply lidocaine and prilocaine cream,
-
impax lidocaine and prilocaine cream,
-
how to apply lidocaine prilocaine cream,
-
how to use lidocaine and prilocaine gel manforce,
Circumcision
Lidocaine/prilocaine eutectic mixture is to be use during circumcision in newborn boys
and is consider efficacious and safe to lessen pain from circumcision.
The European Medicines Agency concludes in its latest (2014) statement on Emla: “Safety and efficacy for the use of EMLA on genital skin and
genital mucosa is not establish in children younger than 12 years. Available pediatric data do not demonstrate adequate efficacy for circumcision.”
They state the cream must be at least one hour before the start of a routine procedure and for two hours before the start of a painful procedure.
Tempe
The spray is a combination of local anesthetics lidocaine and prilocaine in a metered-dose aerosol that is spray directly on the penis to numb sensations.
It is develope by the same group that invente the erectile dysfunction drug sildenafil.
The drug was approve in Europe and is release in the UK market in November 2016 and within the EU will be out for market by, early in 2017
In the United States, the Food and Drug Administration (FDA) is expecte to approve the drug in 2018.
Compendial status | lidocaine and prilocaine cream
- United States Pharmacopeia





Reviews
There are no reviews yet.